 
Thus, by looking at an IR absorption spectrum, a chemist can make some important determinations about a molecule’s chemical structure. Atomic Absorption Spectrum The spectrum of certain absorbed wavelengths of light corresponding to an atom's spectrum of emitted frequencies of light. The discontinuous line spectra of light produced when excited atoms return to their ground state and emit photons of a certain. However, they can't remain at excited state so they drop to lower levels and emit energy. The emission spectrum of a chemical element is the pattern of wavelengths obtained when the element is subjected to a specific excitation. 
Because each of these bonds is different, each will vibrate in a different way, and absorb IR radiation of different wavelengths. The discontinuous line spectra of light produced when excited atoms return to their ground state and emit photons of a certain frequency. Explain the emission spectra When energy is supplied to an atom, electrons gain energy and jump to higher energy levels from ground state to an excited state. For example, a chemist might learn from an IR spectrum that a molecule contains carbon-carbon single bonds, carbon-carbon double bonds, carbon-nitrogen single bonds, carbon-oxygen double bonds, to name but a few. 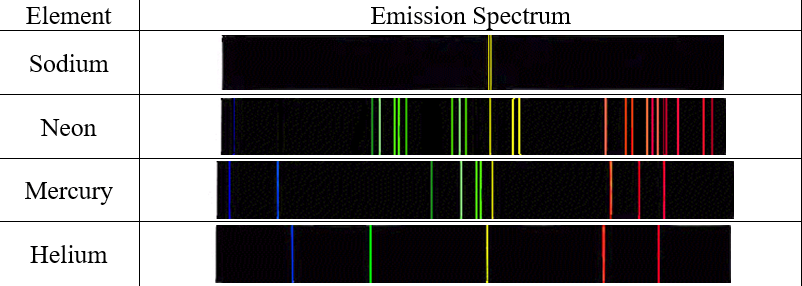
By using special equipment in the lab, chemists can look at the IR absorption spectrum for a particular molecule, and can then use that spectrum to determine what types of chemical bonds are present in the molecule. Just as the energy required to excite an electron in a particular atom is fixed, the energy required to change the vibration of a particular chemical bond is also fixed. Where all wavelengths and frequencies are represented (rainbow) The emitted light corresponds to. This type of radiation is usually not energetic enough to excite electrons, but it will cause the chemical bonds within molecules to vibrate in different ways. However, lower energy radiation in the infrared (IR) region of the spectrum can also produce changes within atoms and molecules. Assign these wavelength s to transitions in the hydrogen atom. So far, we have been talking about electronic transitions, which occur when photons in the UV-visible range of the spectrum are absorbed by atoms. Find step-by-step Chemistry solutions and your answer to the following textbook question: An atomic emission spectrum of hydrogen shows three wavelengths: 1875 nm, 1282 nm, and 1093 nm. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
